In the rapidly evolving world of biomedical science, mRNA vaccine technology is advancing beyond its early pandemic successes with groundbreaking computational and biological innovations that promise even faster, more precise vaccine development for infectious diseases and therapeutic applications. Since the initial global rollout of mRNA vaccines during the COVID-19 pandemic, researchers have increasingly focused on leveraging high-resolution modeling techniques to understand and optimize the structure, design, and delivery of mRNA molecules at unprecedented levels of detail. Scientists have recognized that these computational and biological innovations can dramatically shorten development timelines and improve clinical outcomes for future vaccines, including those targeting challenging diseases such as HIV, influenza, and cancer.
The major advantage of mRNA-based vaccines lies in their modular nature — they use synthetic messenger RNA sequences that instruct cells to produce specific proteins that trigger an immune response. This means that once a pathogen’s genetic blueprint is known, mRNA sequences can be designed and manufactured much more swiftly than traditional vaccines that depend on growing pathogens in culture or adapting weakened viruses. Researchers have now begun harnessing computational design platforms and deep-learning models that can analyze and optimize entire mRNA sequences at high resolution, including untranslated regions and structural motifs that affect stability, translation efficiency, and immune activation. These modeling techniques can process complex sequence information faster and more efficiently than traditional design approaches, enabling more effective candidates to be selected earlier in the pipeline.
One cutting-edge example is the development of hybrid foundation models like Helix-mRNA, which merges structured biological knowledge with advanced attention-based algorithms to evaluate both coding and non-coding regions of mRNA. This type of high-resolution modeling can simulate how different sequence features influence protein production, immune responses, and molecular stability — all without requiring months of standard trial-and-error experimentation in the lab. Innovations like these not only speed up vaccine candidate selection but also reduce costs and dependency on experimental screening during early development stages.
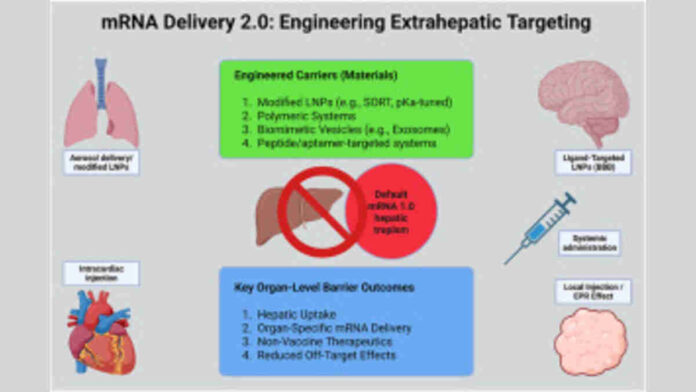
Beyond sequence optimization, scientists are also improving delivery methods and biological design features that affect an mRNA vaccine’s effectiveness. Research into next-generation lipid nanoparticles (LNPs) and other nanoparticle platforms aims to deliver mRNA more precisely to target tissues, including extrahepatic organs such as lungs and immune tissues, which broadens the range of diseases researchers can tackle with mRNA therapeutics. This integration of engineering and biological insight is a key part of building vaccines that work faster and more consistently across diverse patient populations.
These advances play out in real world applications too. For instance, next-generation mRNA vaccine candidates are being tested in clinical settings not only for infectious diseases but also for personalized cancer immunotherapy. A recent study on individualized mRNA vaccines against triple-negative breast cancer demonstrated that personalized approaches could evoke durable T-cell immunity, hinting at a future where vaccines are custom-designed for specific patients and disease profiles. Such targeted precision would have been almost unthinkable before the advent of high-resolution mRNA modeling and computational design tools.
The natural result of this innovation pipeline is that mRNA vaccine development is no longer confined to pandemic response. Hundreds of clinical trials and research efforts are now exploring mRNA vaccines for influenza, RSV, HIV, tuberculosis, and even personalized tumor-specific vaccines — reflecting confidence that the platform’s flexibility and speed can be translated across a wide array of health challenges.
At the same time, regulatory bodies worldwide continue to adjust frameworks to keep pace with this technology’s evolution, ensuring safety and efficacy while allowing rapid approval where evidence supports it. For example, new combination mRNA vaccines, such as Moderna’s COVID-19 and influenza single shot, have moved forward in European regulatory processes, further demonstrating industry and regulators embracing this increasingly mainstream approach to immunization.
In sum, high-resolution mRNA modeling represents one of the most promising frontiers in vaccine science, harnessing the power of computational biology to accelerate development, improve design precision, and expand the reach of mRNA vaccines beyond traditional infectious disease prevention. As researchers continue to refine these tools, the era of fast, adaptable, and highly effective vaccines — once a distant goal — is rapidly becoming a scientific reality.