Pharmaceutical giant Pfizer has moved a major step closer to transforming cancer treatment by advancing its mRNA-based cancer vaccine into late-stage clinical trials. The development marks a critical milestone in the company’s long-term strategy to expand the use of mRNA technology beyond infectious diseases and into personalized oncology treatments.
The vaccine is designed to train the immune system to recognize and attack cancer cells by targeting tumor-specific antigens. Unlike traditional cancer vaccines, which often take a one-size-fits-all approach, Pfizer’s mRNA cancer vaccine is built around precision medicine. Each formulation is tailored using genetic information from a patient’s tumor, allowing the immune system to identify cancer cells more accurately while sparing healthy tissue.
Late-stage trials indicate that earlier phases delivered encouraging results in both safety and immune response. In mid-stage studies, patients receiving the mRNA vaccine alongside existing cancer therapies showed improved immune activation and a reduced risk of tumor recurrence. These results gave Pfizer the confidence to expand testing to larger and more diverse patient groups, a necessary step before seeking regulatory approval.
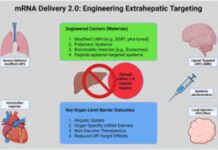
Pfizer’s vaccine leverages the same mRNA platform that proved effective during the global COVID-19 response, but the oncology application is far more complex. Cancer cells mutate rapidly and often evade immune detection. By encoding multiple tumor-specific markers into a single mRNA sequence, the vaccine aims to stay ahead of cancer’s ability to adapt. This multi-target approach could significantly improve long-term treatment outcomes.
The late-stage trials are currently focused on high-risk cancers with limited treatment options, including melanoma and certain solid tumors. Patients enrolled in the study receive the vaccine after surgery or chemotherapy, with the goal of preventing relapse. This post-treatment strategy positions the vaccine as a complementary therapy rather than a replacement for existing cancer treatments.
One of the most promising aspects of the mRNA cancer vaccine is its potential scalability. Once approved, mRNA manufacturing allows for faster production compared to traditional biologics. Pfizer has already invested heavily in expanding its mRNA manufacturing capacity, enabling rapid customization for individual patients while maintaining consistent quality standards. This infrastructure gives the company a strong advantage as personalized medicine becomes more mainstream.
Industry experts see Pfizer’s progress as part of a broader shift in cancer care. Immunotherapy has already reshaped oncology through treatments like checkpoint inhibitors, but vaccines offer a proactive approach by strengthening the body’s natural defenses. If successful, mRNA cancer vaccines could reduce reliance on aggressive treatments such as chemotherapy, improving patient quality of life and long-term survival rates.
Regulatory agencies are closely monitoring the trials, given the novelty of personalized cancer vaccines. Pfizer is working under strict clinical and ethical guidelines, ensuring patient safety and data transparency. Late-stage trials will also provide crucial insights into long-term efficacy, side effects, and durability of immune response — factors that regulators will heavily weigh during approval reviews.
The commercial implications are significant. Cancer remains one of the world’s most expensive and challenging health burdens, and successful mRNA vaccines could open a multi-billion-dollar market. For Pfizer, this innovation helps diversify revenue streams beyond pandemic-era products while reinforcing its position as a leader in next-generation therapeutics.
Challenges remain, including high development costs, complex logistics, and ensuring equitable access once the vaccine reaches the market. Personalized treatments often face pricing and distribution hurdles, especially in lower-income regions. Pfizer has indicated that it is exploring partnerships and tiered pricing models to address global accessibility concerns.
By advancing its mRNA cancer vaccine into late-stage trials, Pfizer is signaling confidence in a future where cancer treatment is more targeted, adaptive, and immune-driven. If the trials succeed, this breakthrough could redefine oncology care and mark one of the most significant medical advancements of the decade.